 
Figure 10.5 illustrates these different molecular forces. 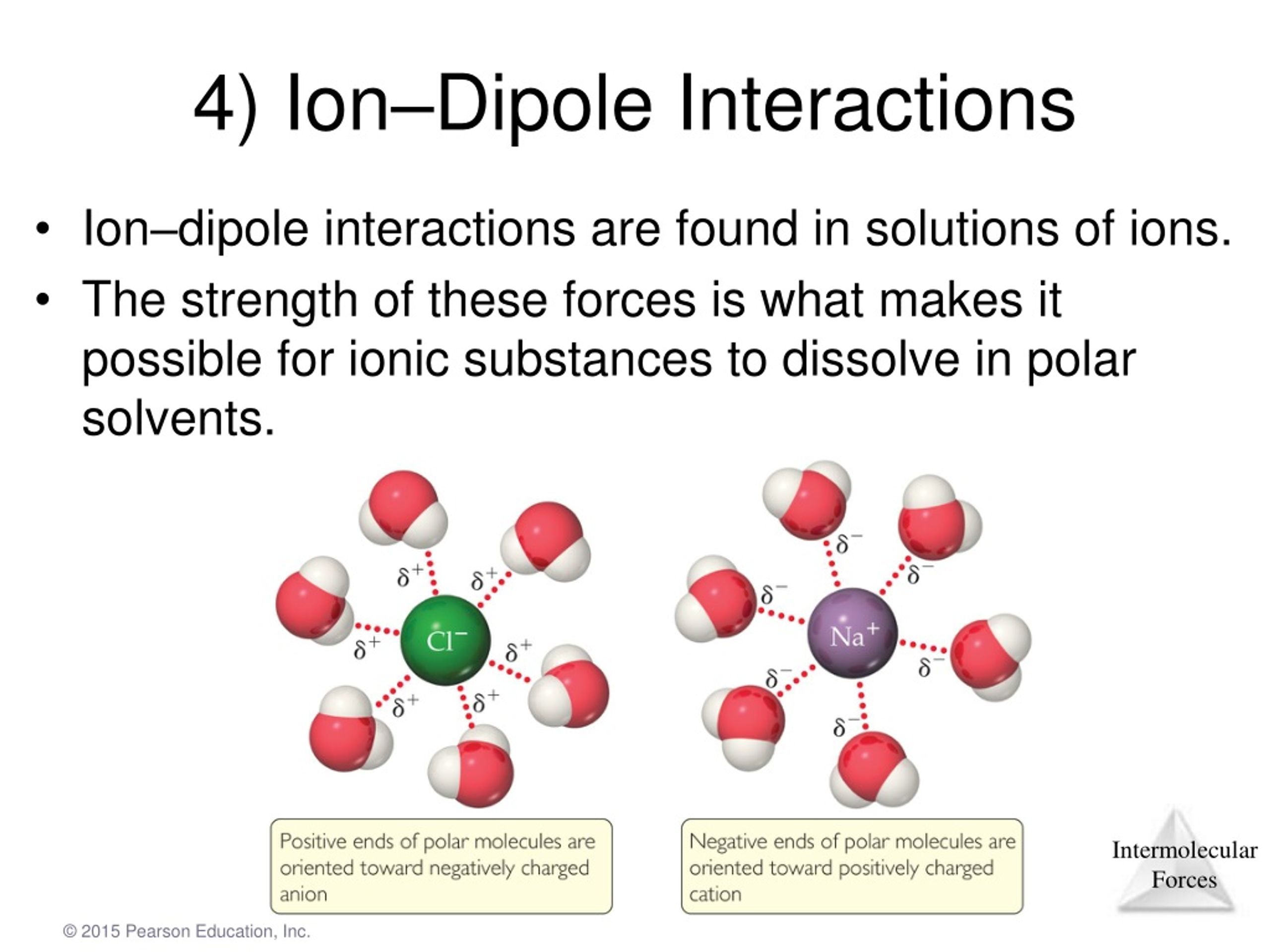
Determine the sites or gaps created when packing (tetrahedral hollows, octahedral hollows). Use Fajans rules in relative polar prediction. Inter molecular forces are the attractions between molecules, which determine many of the physical properties of a substance. Waals, dipole ion, dipole-dipole, induced ion-dipole, induced dipole-dipole, induced dipole-induced dipole, hydrogen bonds). These forces form when ions and polar molecules get close to each other. These forces are by far the strongest intermolecular forces, and their strength can often surpass the strength of some weak covalent bonds. 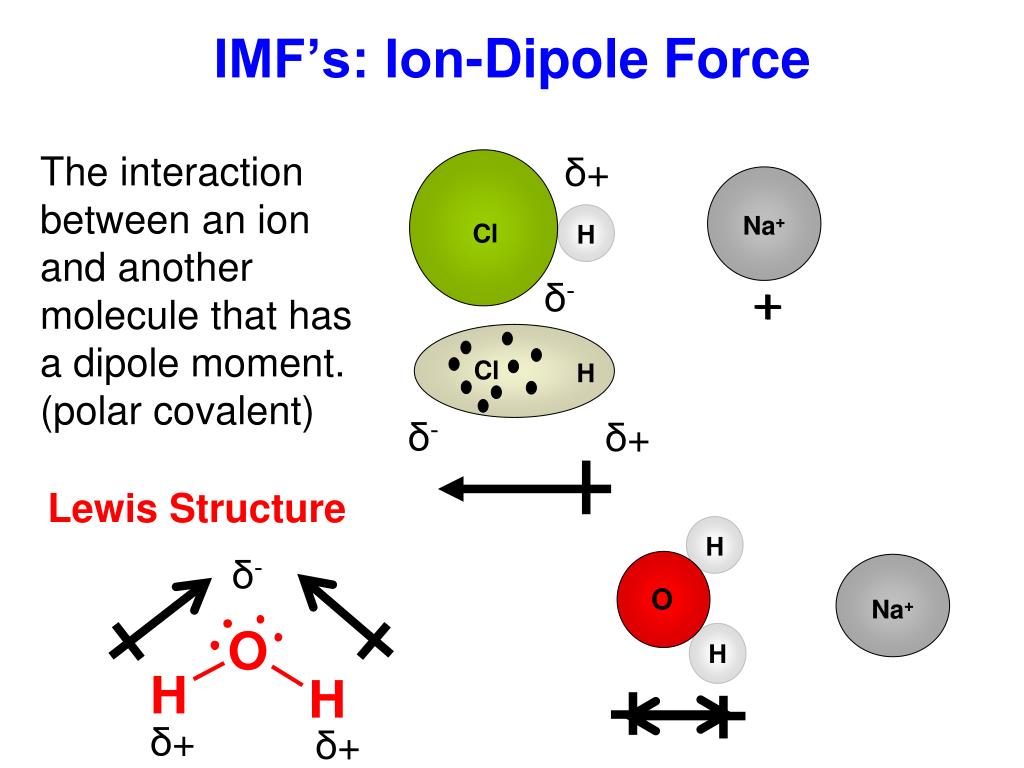
Consequently, in accordance with Equation 4.1.1, much more energy is released when 1 mol of gaseous Li F − ion pairs is formed (−891 kJ/mol) than when 1 mol of gaseous Na Cl − ion pairs is formed (−589 kJ/mol).Ĭalculate the amount of energy released when 1 mol of gaseous MgO ion pairs is formed from the separated ions. Intra molecular forces are those within the molecule that keep the molecule together, for example, the bonds between the atoms. In a mixture of ions and polar molecules, there will be London forces, but also ion/dipole forces. This interaction is temporary because it can be easily overcome by thermal agitation at. Na coming from table salt, NaCl, that dissolves in water) and a polar molecule (e.g. The first two are often described collectively as van der Waals forces. The main force at play here, the ionic bonding, is permanent an ion-dipole force is a temporary interaction between an ion (e.g. The ends of the dipoles in an atom generally possess a small partial charge. There are two additional types of electrostatic interaction that you are already familiar with: the ionion interactions that are responsible for ionic bonding, and the iondipole interactions that occur when ionic substances dissolve in a polar substance such as water. \( \newcommand/mol\right )=-891\ kJ/mol \) īecause Li and F − are smaller than Na and Cl − (see Figure 3.2.7 ), the internuclear distance in LiF is shorter than in NaCl. Hydrogen bonding Iondipole forces and ioninduced dipole forces Van der Waals forces Keesom force, Debye force, and London dispersion force. The molecules possessing permanent dipole show the dipole-dipole type of force. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
